How to CE Mark a Product
14-10-2020 Law & Legislation
As we specialise in creating user manuals for products, many of our clients are involved in the certification process.
As I think it is useful for companies selling any kinds of products on the European market, to have more insight in the certification process and how to CE mark a product, we will talk about it in this podcast episode.
In this podcast, I am interviewing Paul Hoogerkamp, who specialises in assisting companies with the conformity assessment procedures of all kinds of products.
Listen to the interview below and make sure to subscribe to the INSTRKTIV’S INSANE INSTRUCTIONS SHOW on your favourite podcasting platform. You can find the free transcript of this episode on this page as well.
SOME TOPICS WE COVERED:
- When do you need a CE mark
- CE Directives vs. non-CE Directives
- Responsibilities of importers and manufacturers
- Brexit and CE marking
- CE marking of components and parts
- Self-certification
- The six steps of the CE marking process
DO YOU WANT A USER MANUAL TEMPLATE THAT ALREADY CONTAINS THE LEGAL PARTS?
Take the shortest way to a compliant manual. We have developed user manual templates for medical devices (EU and US), machinery and electrical equipment that contain all legal content.
Table of contents
What is a CE Mark?
When do you need a CE mark for your products?
How to identify if your product falls under the scope of CE marking
Difference between CE directives and non-CE directives?
The Declaration of Conformity
CE marking as part of the design process
What are your responsibilities when you import a product?
BREXIT and CE marking
How to CE mark product components and parts?
Can you self-certify your product?
When do you need a Notified Body?
What does the CE marking process look like?
Step 1 of CE marking a product: Identifying the applicable legislation
Where to download directives and standards?
Step 2 of CE marking products: Identifying the applicable requirements
Step 3 of CE marking products: Identifying the applicable legislation
Step 4 of CE marking products: Testing your product
Step 5 of CE marking products: Compiling the technical file
Step 6 of CE marking products: Place the CE mark and sign the declaration of conformity
Ferry: Hi there, and welcome to the show. In this podcast, we're going to talk about how to CE mark a product. Products can include toys, machinery, personal protective equipment, pressure equipment, medical devices, et cetera. As we, as a company, specialize in creating manuals or IFU for medical devices, electrical equipment and machinery, most of our clients are involved in the certification process of their products. With the user instructions that we create, we help to provide clear information on how to use a product, which is necessary for using a product safely, efficiently, and effectively. The user manual plays an important role in the certification process.
One of the purposes of the manual is to warn the user against certain product risks, product safety legislation, in most cases, also gives requirements on the contents of the user manual. What should be in the manual also depends on the product group. The requirements on the user manual for machinery differ from those of a toy, personal protective equipment, measuring device, construction product, or medical device. As I think it's useful for companies selling products on the European market to have more insight in the CE marking process, we will talk about it in this podcast episode. Together with my guest, I hope to provide practical tips on how to go through the CE marking process and self-certify your product.
In this podcast, we will discuss what CE marking stands for on products, when do you need a CE mark for your products, the process of CE-marking in general, and the six steps to CE marking, how you can get or obtain a CE mark for your product, how to identify if your product falls under the scope of CE marking, what are CE marking requirements, and how to check if a CE mark is valid. [music]
My guest for today is Paul Hoogerkamp. He's one of the most well-known experts in the field of CE marking and machinery safety. He's owner of MECID, with which Paul advises on CE marking and product safety. He developed several CE marking-related tools. He's author of several books on machinery safety and risk assessment. He's trainer at the Dutch Standardization Institute, and chairman of several standardization committees such as the Dutch Machinery Committee. Welcome, Paul.
Paul: Okay. Welcome.
Ferry: That's such an impressive list. Every time I speak to you, new things are going on. What committees are you currently involved in as a chairman or a member?
Paul: The current committee is on the performance level. It's known as the 13849 and the 62061 committee. There's also the risk assessment and ergonomics committee where I attend as a member.
What is a CE Mark?
Ferry: Let's talk about CE marking in general, first. Can you explain to us what the CE mark is?
Paul: A CE mark is nothing more or nothing less, that the manufacturer puts a label on it and I says, "This machine or product complies with the product legislation or applicable product legislation."
Ferry: That is European product legislation? When you see a CE mark, you know for sure that the manufacturer of a product did everything to comply with the European directives and the requirements in those directives. Is that correct?
Paul: Oh well, [chuckles] that should be correct, but it isn't because many manufacturers, in fact, are doing their best to do the CE marking in the correct way. There is always some, let's say, minor CE and you cannot distinguish it from outside. We have also, in our countries or member states, let's say, the so-called mark surveillance that is the authority that's looking at bad products. You cannot avoid that the CE marking, sometimes, is not telling the truth, sorry to say that.
Ferry: One of the things that I hear every now and then, and I think it's a misconception, but that people think that the CE mark means that the product is manufactured in the European Union.
Paul: Indeed it could be a misconception. Normally, it's the manufacturer that puts the CE mark on it. CE mark, by the way, it's only an idea from the European Union - not a bad idea - to minimize the so-called trade barriers. That means that the trade barriers are now at the member states, not anymore. You can sell the products everywhere in Europe. Based on this principle, you have to CE-mark a product from the beginning, before entering the market. The CE mark could be that it is stamped in the US, or Canada, or New Zealand, or Australia, or even China, whatever, but when it's entering the European Union, the CE mark should be correct. That means that then, not the manufacturer in the first line is the one who is at stake, but is also the importer. The importer is then the important one.
Ferry: Is it a mandatory thing to CE-mark?
Paul: Yes, mandatory.

The CE Mark
What products need CE marking?
Ferry: To which products does it apply?
Paul: It's a lot of products. The products where there’s legislation present, let's say, if there is no legislation on the subject or products, there is no CE mark, maybe there is another marking or something. In general, if the product complies to the product regulation parts, then, of course, the CE marking is a valid marking.
Ferry: For example, sunglasses fall under personal protective equipment. The PPE directive is a CE marking directive, is that correct?
Paul: That's correct. Sunglasses, it depends on what the manufacturer thinks. You have sunglasses, they are for fun and nothing to help, only to look better, for instance. Now, those sunglasses are nothing. You can only think about it as a general product safety device, so the sunglasses should be conforming to the product legislation that there is no poisonous substances in it and so on. When the sunglasses are under the PPE, that means they have guarded protection for eyes. Then, of course, you have to have a CE marking on it, a small manual on how to do, and to deal with it of course, and the manufacturer has to do some testing so that it can see that a product performs.
Ferry: Let's stick to these sunglasses because you're saying when it doesn't have a protective function, then it doesn't fall under the PPE directive and it's not part of CE marking. For example, when the sunglasses don't have a protective function, but they are intended for children, for example, I think then the Toy Directive applies, which is another CE directive.
Paul: Yes, correct. The Toys Directive. It is fairly simple: if a product falls under a directive, also the manufacturer needs to address this, then, pick out the directive, the product fits, let's say, or is suited. That means that the manufacturer has to decide where the product is intended for, the use, the group, or whatever,... people. If the manufacturer says that the intention is only for, for instance, children, toys, if it is intended for PPE, PPE, it could be intended for both. It depends on what the manufacturer thinks - I'll give you an example of this problem, what is more technical than juridical. If you have, for instance, dolls, if you have a Barbie doll. The Barbie doll is, of course, for playing. You also have Barbie dolls for exhibitions. They have clothing, in a variety of clothing, and they are only for showing off. They are clearly intended not for playing. They are not sold in the shops. That is clearly a non-toys directive issue.
How to identify if your product falls under the scope of CE marking
Ferry: For example, machinery, there is Machinery Directive which is a CE directive and applies to all kinds of machinery. When a machinery, for example, is intended for artists to use in performances, then it's excluded from the Machinery Directive meaning that you always need to consult a directive to see if your product is included or maybe it's excluded by that same directive.
Paul: That is correct. You have to check. One of the obligations of the manufacturer is to check if your product falls in one or two or maybe three or four, whatever directives. That is the product directives. You can find them on the internet: EU Growth. There you have an overview of product directives. Then, for the manufacturer, it's a job. Check if your product falls under the definitions of this (directive), and if yes, then you have to comply with that.
Ferry: I'll include the link that you mentioned in the transcriptions of this podcast, but you're saying so on that page of the European Union, there's a list of European directives.
Paul: That's correct. There's a listing because you have to scroll down sometimes, and when I [chuckles] explain to the person from where you can find it, they say, "Oh, there was nothing." Scroll a little bit downwards, and you have a list of directives. It's a list box. There you can click on the directives. Probably more than 60 for those directives dependent on the area you want. For machinery, of course, machinery directive, but also dependent on the part of the machinery, there could be more directives applicable at the same time.
Ferry: It's not always one directive that may apply to your product.
Paul: Yes, that's correct.
Difference between CE directives and non-CE directives?
Ferry: You're saying there are maybe over 60 directives and regulations listed there. Are these all CE directives?
Paul: No, but they are New Approach directives. There are more directives that are not CE directive related. For instance, if you have transportable pressure equipment, which are those famous bottles for helium, oxygen, nitrogen, and so on, they have a so-called P mark. That is the P from the circle, P3.14. That marking is available for those transportable pressure equipment. You have also the WEEE, that means Waste Electronic and Electrical equipment, and therefore, you have mark with this bin-- It's not CE, but it's a bin. You have several markings.
The WEEE symbol
Ferry: Why does the European Union make a difference between CE directives and regulations and non-CE directives or regulations?
Paul: There could be several reasons, political, technical, I don't know. It's depending on the subject and what is the need for the regulation of the sectors. In fact, it started in 1980, 1985 with this new approach directives. They thought, "Well, we have to get rid of these trade barriers in the member states." When you get rid of it, what to do? What kind of legislation to apply? For us, it started with the Low Voltage Directive. Later on, they said, "Well, if we have some new approach directive on a general level, maybe we can have a several, let's say, documents." One of them is the machinery directive, the PPE directive, and so on. If there's a need for a sector, they can do it, but it depends. There are sectors that are not regulated directly. Most of the time, there are indirect or local regulations. That means local member states regulations are then there. Most of the legislation now either is in the member states, or have more related higher-level legislation that is coming straight from, let's say, Brussels or at least Europe.
Ferry: Actually, you're saying that how this has evolved, it has a historical background. In the '80s, when they started with CE marking, the European Union or European Commission, decided that certain products need to be regulated a bit more than other products. They started with certain product groups in order to get rid of trade barriers.
Paul: Yes. It started from industry. There was a huge lobby from industry. There are two names I am familiar with regarding the lobby. There's Pehr Gyllenhammar from Volvo, from Sweden, and Wisse Decker from Philips in the Netherlands. They wholeheartedly said, "We need to have regulations to get rid of these barrier trades." The idea behind it is quite simple. When I have a machine, no let's say a drilling machine, and I have to comply with the Dutch legislation, at that time, I have to go through the KEMA, for instance, and they certified my machine. When I went to Germany, they said, "No, KEMA, that’s not okay. We have another marking or Geprüfte Sicherheit, the GS marking." At the end, you had a lot of so-called Notified Bodies. A lot of certifications were needed to enter the European market. This costs a lot of money. Well, the testing is not really different in those testing houses or modified bodies. That means that all the testing was a burden. Get rid of that burden if the KEMA has tested it once, why is it not valid for Germany or England or so on? That needs to be addressed, that's why.
Ferry: It sounds like it was almost impossible for small and medium-sized enterprises before CE marking was introduced to sell products Europe-wide.
Paul: Not impossible, but it costs you a lot of money. [laughs] That was a trade barrier, of course. Now, this European model is widespread all over the world.
Ferry: Do you think that this European model has inspired other countries to introduce a similar system?
Paul: The similar system, yes, because I was also involved for the European Union to go abroad to say what it is,.. the Machinery-, EMC-, and all the directives. What you see is they like the idea. Have a high level of regulations, and then give the standardization building bricks, but at a high level, it's clear: the machine should be safe, and the voltage/ low voltage should be safe, et cetera. This high level was already there, and many countries liked that. The European Union has here a unique selling product: legislation.
Ferry: I fully agree. You're mentioning that getting rid of trade barriers is one of the advantages of CE marking. Isn't it also a big advantage that you guarantee a certain level of safety for products when they comply with the requirements?
Paul: Of course. Still, there is a problem: if you apply the CE marking in the right way and do the CE marking process correctly, there’s no problem. You always have a problem with how to check…. Let's say I can put the CE marking stamp on it, and I'll put my product on the market. If the product is really bad and our excellence, of course, I will be swept off the market with the whole company and the people. The problem is how do you check, generally, all products in all situations. That will be an issue.
The CE marking process
The Declaration of Conformity
Ferry: In general, putting the CE mark on the product is an easy thing to do, but we'll talk about that later. It's actually the last step in the CE marking process. You only put the CE mark on the product when you are sure that your product complies with the requirements.
Paul: Yes. There is a funny story about it. In the past, when the machinery directive was applicable in 1992, there was an exhibition on agriculture machinery. At that time, there was a guy, and he said, "Hey, guys", on the booth of the machinery, he said, "You have not CE-marked your machinery. Here is a sticker, put it on." There was a guy with stickers at the exhibition. Afterwards, there was also a labour inspection, and they said, "Well, we have a nice A4 paper and you can cross, CE marking present, yes/no. Let's say manual present, yes/no, and other remarks." We got in a situation where people were putting CE stickers on it, and just 10 minutes later, the labor inspection came and said, "Oh, the CE marking is yes. Do you have the manual?" "Maybe." They cross yes or no, and suddenly, we're going to check next half year if you're okay. This is a hide and seek method. That was in the early beginnings.
Ferry: So meaning only the CE mark was present, the manual wasn't, the risk assessment wasn't, and everything was missing for the rest.
Paul: Yes, of course. Then, what does the CE label mean? I have the declaration of conformity (click on the link for a CE certificate download), it is a piece of paper. You sign it, and number it. No big deal.
Ferry: To summarize, when you sign the declaration of conformity and put the CE marking on your product, you declare that your product meets the requirements as indicated in the European directives.
Paul: That's correct. You're not only responsible, but also accountable if something goes wrong. Your signature means a lot when you have this declaration, and you put your signature on it on behalf of your firm or whatever, then you're liable when something is going wrong. You're responsible, liable, accountable. That means a lot. People were just, "Oh, well what did I sign? Oh yes, there should be a technical file? Wow, what is a technical file?" What we saw in many firms is a rollback of the situation, not getting in advance, what is the legislation and how to implement it in the design process? At the end, put a CE sticker on it? No, it was quite the reverse, you put the CE sticker. "Okay, that means I have to sign something. I sign something, but what does it mean?" Then, it came clear to many people or many persons or many manufacturers, "Wow, there's a lot of more that is here at stake. We have to design safe machines and not just check if the machine is safe, no, in the design it should be safe."

Example of a Declaration of Conformity
CE marking as part of the design process
Ferry: Yes, and then you get to the process then where the CE mark is like an integral part of the design process.
Paul: That's correct. That should be. We started straight from the beginning to tell people, "Please, it's not only a paper. It's a process in the design. You have to make a safe design.
Ferry: The funny thing is actually I studied industrial design engineering at the University of Delft, which is quite technical. You learn how to design products, mostly electrical equipment. During my studies, I never had any college about CE marking. [laughter]
Ferry: People said, "Okay, when you follow standards, when you get involved in the very beginning with everything that is regulated, standards et cetera, it kills the creativity of the design in the design process.” What do you think about that?
Paul: I'm quite the contrary. I think you need the boundaries. You need to know the limits. If you're in the desert, every direction is okay. You're not creative. People think that you'll be creative without any borders or limits. It's wrong. When you have borders, limits, then you can be creative. Know your limits. Know the limits of the legislation. Know the limits of the user, whatever, and then you can be creative.
What are your responsibilities when you import a product?
Ferry: Yes, I agree. Another question that crossed my mind, what we hear as well every now and then is that we get some emails from companies that are importing a product from China, for example. They ask us to do the user manual. I ask them, "Which directives apply to the product?" Then, they say, "I have a declaration of conformity." They say, "It's signed by our Chinese manufacturer, do we need to put it on our name?" [chuckles] What you know then for sure actually is that they have no clue about what responsibilities they have.
Paul: Yes, and the responsibilities of also the importer, because from China, there's a problem, you cannot go to China and say, "Hey, this is wrong," and have them for court here in Europe. That's not done in our present times. The one who imports this stuff, product, whatever, he is liable, accountable, responsible whatever for this product. Of course, in China, you can make a good product, no problem, but the importer is the one who is responsible for that. Chinese manufacturers, CE signed by whatever P. Chin or whatever, no problem, but there is an importer or a responsible person in Europe. They have a technical file. Like, for instance, a technical file in English or another, let's say member state language.
The EU Member States
Ferry: The importer is considered to be the manufacturer.
Paul: Yes, de facto, they call it a manufacturer. De facto, they have the same obligations. You can also find it in decision from the European Commission, how to deal with it and what are the obligations of an importer, of a distributor and a manufacturer.
Ferry: The answer to the question, who should CE mark a product, is?
Paul: In fact, the manufacturer is the basis. Wherever he is or she is on earth, but then if it's in Europe, it's of course, the manufacturer in Europe, but it is outside Europe, then there is something like an importer or a responsible person that has to sign with it or at least is accountable for the technical file and so on.
Ferry: Yes, this responsible person is called the authorized representative.
Paul: Authorized representative, correct.
BREXIT and CE marking
Ferry: What's the case with the UK at the moment? When I import something from the UK, that's from outside the European Union, right?
Paul: Yes. Outside the European Union, but not outside the European economic area. They are still in the economical area unless they make a so-called hard Brexit whatever it is. If they do it, then maybe they become some bounded third country that they call it. Basically, they use the term third country that's from outside Europe. They are not in the European Union. What are they going to do? They are going to the EFTA or going somewhere else, I don't know for sure, don't bet on this. When a product is from England to Europe, of course then the technical file should be on the European member states grounds. It's interesting because the notified bodies, they're losing their number. The notified body in England makes a certification, it's not valid anymore. Let's say product troubles are with Brexit incorporated, let's say.
Ferry: We'll need to see how it evolves. That's the UK, what about France? For example, when I import a product from France, a country within the European Union, then it's not seen as an import anymore? It's trade within the European Union? Actually, my question is who is seen as the manufacturer when I import a product from France or from Ireland and I sell it on the Dutch market or German market?
Paul: If the product was produced in the Netherlands, it's equal if it's produced in Ireland, France, or Germany. The trade barrier is not anymore there. We are one big country for this part regarding product safety, we are one together.
Ferry: Am I not seen as the distributor when I import a French car and I sell it on the Dutch market under the French brands?
Paul: For cars, of course, there are automotive directives and so on, it's not only machinery. The same regulation is applicable. When it's made in France, you can sell it on the European market, that means the Netherlands, that means Germany. It's no trade barrier, in fact, but the product should be then regulated. If there's a non-regulated product of course, then you can face some trade barriers.
Ferry: Okay. Let's say I'm importing a product from outside the European Union, from China, the Chinese manufacturer already placed the CE mark on the product, I am the importer, how do I check if a CE mark is valid?
Paul: If the CE mark is correctly laid out, well that's in the directive, then, of course, the CE marking is a valid mark. Then, you have to check several other situations. Maybe you can check against some product standards. Maybe it is valid that you can check it against the legislation in general. It depends on the product.
How to CE mark product components and parts?
Ferry: Yes, it depends on the product, it can be a toy, for example, that I import but what happens as well is that companies import product parts that they use in machinery or that they use in electrical equipment. What do I need to do, for example, when I import a small electronic component that I use in my electrical device?
Paul: Still, the manufacturer, the end manufacturer is responsible for his product. That is the same question all the time. If you pick parts out of China or out of everywhere, then the parts-- It depends on your purchasing department, or let's say a technical testing department, if you have the right component. You import a resistor, a tiny, electronic component, now if the resistor is okay, no problem. If the resistor can burn by mistake and component failure, who is responsible? Of course, the manufacturer who assembles this within this resistor.
Ferry: Let's say that I'm a manufacturer based in the European Union and I want to purchase some electrical components, would be the first step that I source a Chinese supplier that at least says so that they can deliver components that comply with the European directives?
Paul: The problem is that the components themselves cannot directly comply with the directive or technical values. Of course, electrical components do have a compliance within the RoHS directive. That means that there are no, let's say foreign substances in it or substances that are not allowed. If the substance is okay, and let's say talk about a resistor or a capacitor, it's okay, then you can use it. There is no CE on it, but you're depending on the buy-in stuff, the testing, and to check if the RoHS compliance is okay.
Ferry: Sorry to interrupt you, Paul, but for example when I import a cable. Cables quite often have to comply with the EMC directive or even a Low Voltage Directive.
Paul: EMC, depends, but low voltage can for sure, if it's above 50 volts. Depending on the cable issue, we have the Radio Equipment Directive, it could be from zero Volts. Yes, for cables, but there is a standard also for the cables.
Ferry: Then, I'm an European manufacturer, I'm importing cables from outside the European Union, and I want to make sure that the cables that I purchase, that they already comply with the European requirements.
Paul: You can do two things in this way. You can check that the Chinese are complying. You have to check. You have to have quality assurance and so on. You check if they comply. If they comply, no big deal. They can print a CE mark logo on it. If the cable is not marked anyhow, then you should be aware as a manufacturer, "Be prepared: this is a non-marked issue." It could be that the specification is okay, but why should you go along with non-marked cables and give yourself the extra responsibility for that?
Can you self-certify your product?
Ferry: Exactly. Another question regarding the CE marking. You often hear that CE marking is self-certification. Is that correct?
Paul: Yes, but the terminology is a little bit decorated. Of course in the design process, let's say almost every machine or every product so to speak has to be designed by a manufacturer. You do it by yourself. That means that when there is not a notified body involved at the end or somewhere halfway, dependent on the product, you have to do it yourself. The best way is to do it all the way yourself because you're responsible in the end, not a notified body, not a third party. You're responsible as a manufacturer. You have to do it yourself. The self-certification was a little bit a terminology of when you have a notified body involved, then the certification is valid, and self-certification is not valid. Self-certification is like trusting on blue eyes. In the USA, we have the UL certification. That's a third party. There's always a third party with the product involved. With self-certification, no third party is involved. That's the idea in Europe, to check afterwards if the product is okay, by a market surveillance authority. But you can also do it before it's entering the market. It depends what you choose. It depends how many authority you have to deal with it.
Ferry: Actually, you're saying that CE marking is always self-certification. In some cases, you need additional notified body testing.
Paul: Yes, there are some cases that the notified body involvement is very intense, so, it's not at the end a type test, for instance, but it could also be a design check. For instance, for lifts and the cabins of lifts, the notified body is also checking your quality system, for instance, and also the design drawings. That can be quite deep, this involvement.
When do you need a Notified Body?
Ferry: Can you explain a bit more what a notified body is and when you need a notified body?
Paul: It is in the directive. If we take the machinery directive, as an example, there is an Annex IV. They tell in Annex IV which machine, related parts or whatever,...In the annex itself there are appointed several machines that need the assistance or at least involvement of a notified body. It is a list, but let's be honest, it's 95% of all machinery is self-certification. In this Annex IV, are only a few product-related machinery, like a circular saw, like a press brake, with hand input.
Ferry: We did a podcast episode about medical devices a few weeks ago. For example, we discussed that class I medical devices don't require notified body testing, but other classes do require NoBo testing.
Paul: The manufacturer is in fact the person, the one who says, "This machine is complying to the directive or directives." Not a notified body. They are more or less involved to see, for instance, noise emission measurements, that the measurement is done okay and that the measurement is done in the same way so that the measurement is reproducible in Germany, in France, and so on, but the notified body keeps that level of confidence in Europe.
Ferry: Is a notified body the same as a testing institute or is there a difference between a testing institute and NoBo?
Paul: Yes, it could be that the NoBo can do testing, but testing is not required to be done by a NoBo, it depends. Testing, if you have test equipment, you can be a testing institute because you know how to test and how to test on EMC, low voltage and so on. You can have this, as a third party, give test results that are independent. Test is okay, but a notified body, if they do tests, then the test is also done in the same way, let's say as a test house, but they can have this responsibility, what is also in the regulation and obligation, in the regulation.
Ferry: For example TÜV. Is TÜV a notified body in certain areas?
Paul: Yes, a notified body in certain areas.
Ferry: SGS?
Paul: Yes, but you can look it up on the European site. The site that's called Nando*, but that is a site for notified bodies that are recognized in Europe. Also, there are recognitions in America, Australia, and so on, but those recognitions are bound to a certain product, like EMC testing is okay, but certain products they can also check or test.
*Nando provides an overview of Notified Bodies and is neither a CE mark database or CE mark directory
Ferry: Let's say I have a certain product, then if TÜV or SGS is accredited to be a notified body for that product, they can be the notified body for that product. They can do certain testing or the process checks for that product. I can also go to them and approach them to be not as a notified body, but just to do, for example, EMC testing or LVD testing.
Paul: Yes, it's correct.
What does the CE marking process look like?
Ferry: Okay, thanks. Paul, that's a lot of information about CE in general. What I like about CE marking is the structured process. It looks really complicated, but the process to get to CE marking, you can boil it down to six steps for all kinds of products, which makes it, I don't want to say easy but at least it's structured. Before we dive into the details, can you describe briefly how the CE marking process looks like?
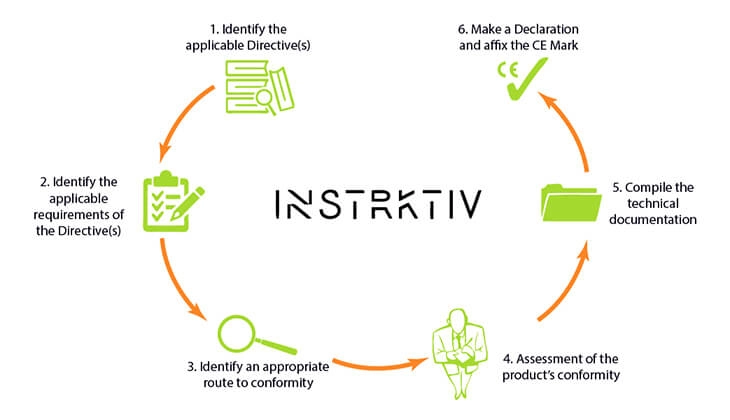
Paul: You mentioned the six steps. Of course, it looks easy, and it sounds easy, but sometimes it's not easy. The six steps are generally: step one, where can I find the directives? What are the applicable directives, or let's say applicable legislation and standards? Second is to check the requirements. Third step is to check your conformity. Do you need a notified body, yes or no? Most times no, but okay. Then, you have testing. Then step five, assemble a technical file. Then, the last step is to draw the declaration of conformity and put the CE label on it. That's the six steps. They are also made visible on the EU site, eu.growth, on the CE marking. You can find it as a manufacturer.
Ferry: Those steps apply to all products regardless whether it's a medical device, a toy, a machinery, PPE, et cetera.
Paul: The steps are generally applicable to all products, but dependent on the legislative part or the product, those steps could involve a lot of work sometimes.
Ferry: Let's say I'm a company located within the European Union, and I want to import face masks from China.
Paul: [chuckles] Yes, that's an actual one. If you want to do it, then the facemask needs to be tested by the notified body because they're PPE involved categories and so on.
Step 1 of CE marking a product: Identifying the applicable legislation
Ferry: Let's start at step one. I want to import a facemask. Step one, you said is identifying the applicable directives and standards.
Paul: No, wait a minute. You said import. The manufacturer does do the six steps. The Chinese manufacturer has to check what are the applicable directives in Europe, first. Then, they have to check the requirements for this facemask, in particular: the standards. If they apply this well, they also have a notified body attached to it who is checking that, and then the testing. Now, the importer, if all the steps are done properly, the importer says, "Okay, this one is CE marked, I can import it. Where's the technical file?" and so on. He has seen this, and then he can import it.
Ferry: Now, you're assuming that the Chinese manufacturer started with step one in their design process, but let's say that the product already is manufactured and I approach a Chinese manufacturer, I'm ordering a batch of PPE and I want to sell them on the European market, I need to compile my technical file. Step one is to identify the directives or is this not a good example?
Paul: Yes, you can do it, but the idea is now you reverse engineer the situation.
Ferry: That is what happens in daily practice quite often.
Paul: Correct. I agree. This is quite often, but then if you have to check if a product complies, of course, take step one, what is the directive? This is an easy one. For this facemask, is a PPE. That means point one is done. There's a standard for it. That's no problem, you can find it.
Ferry: There are directives and there are standards. It's clear to me, but maybe some people don't know the difference between standards and directives.
Paul: The directive is, of course, the obligatory part. The standards are more voluntary. Let's say, if you apply the standard correctly, then you have a so-called presumption of conformity. That means that applying the standard the right way, gives you that you comply with the directive.
Ferry: It creates the presumption of conformity?
Paul: Yes, the presumption of conformity, which is a juridical term, and you can discuss lengthy about it, but the idea is that if a standard is technical, let's say it gives you the technical basis on the present state of the art of a facemask, and if you comply to those technical issues in the standards, then you're, let's say, comply to the directive. The standard becomes an important part, but not the end.
Ferry: The personal protective equipment directive applies to all kinds of PPE, and then there is most likely a specific standard for facemasks giving really detailed technical requirements on the design requirements on the product, but also maybe the instructions, the testing, the way the product needs to be tested, et cetera.
Paul: By the way, those standards now, particularly face masks, are free because of the European Union and the COVID problems, they said, "Those standards are now free. From every institute, you can get this for free." It's not normal, but you have to pay for standards because it is a private enterprise. The standard you can see and then is an easy one, if I import facemasks, then I can check against the standards if this facemask is okay.
Where to download directives and standards?
Ferry: They are for free the standards, you're saying? Does it mean that I can download them from the website of the European Union? Or where can I find directives and where can I find standards in general?
Paul: Yes, the directives, you can download, of course, free in the languages of the EU member states. That is on the EU space.growth site. If you dig a little bit deeper, there is also one that is special for this particular PPE during this COVID situation, that those standards are now released from the Standardization Institute as free standards. If you go to the Dutch Standardization Institute or the German and you ask for those specific standards, they only ask your proper email address and so on, but then you can download it for free, but it's only restricted to several standards relating to PPE for this time.
Step 2 of CE marking products: Identifying the applicable requirements
Ferry: Let's say I've downloaded the directives, I've downloaded the standards that apply to my product, this facemask, in this case, what's next? Step two, you mentioned that you have to determine the requirements that apply to the product. That's correct?
Paul: Yes, that's correct. That means that the PPE requirements now, you can take the standards and see what those requirements mean. They are filled-in requirements for a particular case, in this case, this facemask. Your PPE,... general technical regulations, or let's say the annex, you can pinpoint them into the standards and say, "Okay, I understand what I have to do, I have to do some tests for facemask." Then, you have to pick the standard and to find the right requirements, but that is an easy one. Your requirements are there. In our example, we had the reverse engineering. We go down, take the standards and check if the thing is according to standards.
Ferry: Do all the requirements in the standards apply? Or are they all relevant for that product? Or is that something that you need to do as well?
Paul: What you're asking now is the standards way. You check always the scope. That means, if your facemask fits in the scope of this standard, that’s the first check, of course, and if it is, in this case, in this example, then okay, this standard is applicable for face masks. Then, of course, your specific face mask could be a little bit different from the scope. Then, you have to check again to the directive if something should be added or minimized or-- I don't know.
Ferry: Yes, or is obsolete. In those cases, maybe certain sections do not apply.
Paul: Correct.
Ferry: Actually step two is reading and understanding the standards.
Paul: That's correct. It's not reading, it's reading, studying, understanding.
Step 3 of CE marking products: Identifying the applicable legislation
Ferry: [laughs] Okay, cheers. All right. We've determined the requirements. Then, we go to step three, that's determining the route of conformity. Is that correct?
Paul: Yes. That means that certain PPE has to go to a certain notified body and the notified body could be in the EU, the notified body could also be an agency in China that's under the jurisdiction of the EU and there are certain-- For instance, you mentioned earlier the TÜV, SGS, Dekra, name it, each notified body has some agency in Asia and they do the checking under their umbrella.
Ferry: With determining the route to conformity actually is meant that you have to identify if you need notified body testing or if you are allowed to do the testing yourself?
Paul: Yes. The notified bodies, by the way, they have a number, you can find it on the internet, and then you can see if this notified body is the right notified body to do this checks.
Ferry: That's for personal protective equipment, there are certain notified bodies that are allowed to or accredited to do PPE testing?
Paul: Correct. Yes. There are also notified bodies who have no accreditation for those testing. If you have no accreditation, you're not allowed. You can put a number on it, but it is invalid, fraud.
Ferry: Then, notified body testing is one route to conformity. What are other routes to conformity?
Paul: That is the so-called self-certification, but in this case, I don't know for sure if the self-certification is appropriate, but if the self-certification is okay, depending on the facemask and exactly what the mask does. You have FPPE, PPE, and so on, depending on that. If the most stringent regulation is applicable, then you need a notified body. Otherwise, you can do testing or even test at your own premises. That means you can do testing in China, but then, of course, you need the right testing equipment and then you can challenge these batches of facemasks because have they undergone the right process in the manufacturing plant in China? I don't know.
Ferry: Yes, clear. For example, when we're talking about facemasks now, but for example, when we use a toy as an example, that you don't need a notified body. You are allowed to do the testing yourself. You can do it on your own premises, but then you need to have, for example, the right testing equipment to test on flammability or to test its mechanical properties.
Paul: Yes, that's correct. Those testing and the requirements for those testing are in IEC standards, for instance, or ISO standards depending on what testing equipment. The test equipment itself needs to be qualified for that. You can, from a standardization point of view, see a lot of qualification standards for processes, for equipment.
Step 4 of CE marking products: Testing your product
Ferry: We already moved forward to step four, which is the actual testing. How do you know what testing is needed? Is it correct that the directive gives requirements on the products and for certain requirements you need testing in order to prove if your product needs meets that requirement?
Paul: You can easily say each requirement needs some kind of testing. It's visible, it is there,.. a safeguarding, what are tests on safeguarding? Lean on it, it doesn't bend--
Ferry: Sharp edges.
Paul: Yes, for instance, but it is easily tested by, don't do it, but put your finger on it, on a piece of paper or whatever, you can see the sharpness. Of course, this is visible, but does it need the word testing? We think the word testing is then that you have a test brand like a Volt measurement apparatus or DC current measurement or something like that, what you cannot see as physical. Each requirement has some, let's say test, it's there, visible. It's a visible test.
Ferry: For example, for EMC testing, you can follow a standard, you can follow a procedure in order to make sure that your product complies and the test reports, that are the result of that testing, will be part of your technical file which is actually the proof that your product complies to that requirement. Also, like a photo of a product showing that there are no sharp edges, for example, can be also your evidence and be part of your technical file.
Paul: Yes, a photograph of no sharp edges is a little bit off the edge, let's say [chuckles] because you cannot see from a photograph how sharp it is in practice [chuckles] but yes, the idea is correct.
Ferry: Let's take a small step back. When I'm analyzing, studying on the requirements in step two, then actually what I do is per requirement, there are like loads of requirements in the directs, per requirement, I decide whether it is myself or whether it's regulated already, what testing or route to conformity I need, is that correct?
Paul: Yes. Those steps are in the directive, but also in standards are some routes or testing steps to be taken depending on the directive and those standards. It's not generally that I say, one route is okay, there are several.
Step 5 of CE marking products: Compiling the technical file
Ferry: All clear. Then, let's go let's move forward to step five, then I have my testing reports, I have all kinds of evidence, then in step five, I need to compile my technical file.
Paul: Yes, that's a little bit tricky because it's put in step five. If you start at step five to collect the data, and to put in a technical file--
Ferry: You need to go through all the previous steps.
Paul: Yes, step one should be to make a technical file.
Ferry: Yes, exactly.
Paul: Of course, if you identify the applicable regulation or standards, then you put in the technical file, "This is what I found on that. This is machinery because of this." Or, "This product is toys because of this." Your file is starting a little bit earlier than step five.
Ferry: What else should be in your technical file? How do I know what should be in the technical file for my facemask for a toy, for a medical device, construction product, et cetera?
Paul: That's also in the directive. In the directive itself, there are other conformity routes, but also what is needed in the technical file. You make a technical file in order to, let's say, reflect that you've done the right documentation process and the right things to make this thing safe, or making safe CE marking. In fact, the market surveillance authority can ask you to hand over parts or a certain part of the technical file. The technical file is not that you make for others, you do it for yourself and authorities.
The user instructions are part of your technical file. Watch these videos on how to make user manuals for medical devices, machinery or electrical equipment:
Step 6 of CE marking products: Place the CE mark and sign the declaration of conformity
Ferry: All clear, and only when you have your technical file ready, which can be seen as the evidence that your product complies, then in step six, you can put the CE mark on your product and draw off the declaration of conformity?
Paul: Yes, and that is also part of this technical file. To be precise, the parts that are handed over to let's say outside the manufacturer, that is the part of the technical file regarding the manual. You have to have a copy of the technical file and the declaration of conformity, either you have to have a copy, or the declaration conformity put in the manual, depending on the product itself and the CE marking, of course, that is on the product. There are a few items of the technical file that are copied or in the technical file. The technical file is at the premises of the manufacturer, at least in the EU, and let's say has a copy of the technical file.
Ferry: Thank you, Paul. We've discussed so many things, and I hope we proved a clear overview of the process and what you should do to make sure that your product complies. Thanks again for being an awesome guest.
 |
Ferry Vermeulen is a technical communication and compliance expert. He also is a parttime trainer at the Dutch standardisation institute (NEN). Listen to the INSTRKTIV podcast on Spotify or read one of his latest blog articles. Linkedin I Spotify I YouTube I Facebook I Twitter |
DO YOU WANT A USER MANUAL TEMPLATE THAT ALREADY CONTAINS THE LEGAL PARTS?
Take the shortest way to a compliant manual. We have developed user manual templates for medical devices (EU and US), machinery and electrical equipment that contain all legal content.
